Editor’s Note: I first heard about Jane Eliza Stark from Rolfer™ Ron Murray. In looking into her book, Still’s Fascia, and communicating with her, I thought that her work on fascia, its fluidity and contiguity, would be of interest to Rolfers, who have long been interested in osteopathic thought. Here we present an article she wrote, originally published in Thinking: A Review of the Sutherland Cranial Academy of Belgium [Volume 12, January (2011), pp.18-19] and reprinted here (lightly edited and with additional images) with permission. Following this article, we have an interview with Stark conducted by Ron Murray, and then a commentary on and review of her book.
“Popular Ruts” certainly seems like an odd title for an article about fascia, however, the phrase is an apt summary of how fascia is commonly understood today. Ensuring our logic and practice does not fall prey to the popular ruts of our time is fundamental to osteopathy today.
The term ‘popular ruts’ has its origin in the depressions or grooves or ruts created by the wheels of covered wagons (see Figure 1) as they traveled across the sandstone terrain of the American Midwest. As wagon after wagon headed westward during the 1800s, each successive passing of a wheel enlarged the rut. This process continued until the ruts were so unavoidably large that each subsequent wagon wheel was obliged to travel within the confines of the same rut. Some of these depressions were so deep and so permanent that they remain visible today, 150 years later (see Figure 2).

Figure 1. Covered wagon from the 1800s. Photo by Verne Equinox (Own work) [CC BY 3.0 (http://creativecommons. org/licenses/by/3.0)], via Wikimedia Commons.

Figure 2. Large wagon-wheel ruts preserved in sandstone are still visible in the American West. Photo by Ken Lund [CC BY-SA 2.0 (https://creativecommons. org/licenses/by-sa/2.0/legalcode)], via www.flickr.com/photos/kenlund/66695096.
The problem with ruts lies in the fact that, not only are they are easy to fall into, but, more importantly, they are extremely difficult to get out of. Unavoidably drawn into a rut and then trapped there is exactly how A.T. Still viewed the traditional medical teaching of previous millennia. He wrote (Still 1897, 285), “Since the days of Aesculapius [Greek God of medicine and healing] the delusion has flourished that man must swallow medicine to rid himself of disease.” Still accused the medical system – educators, practitioners, and even patients – of using oral medicine to superficially relieve symptoms; they were all, in his opinion, in the same rut . . . a very, very deep one.
The deliverance of a community from entrapment in rut-like thinking requires a paradigm shift. Copernicus did this, displacing in the minds of Earth’s inhabitants their unquestionable belief that planet Earth was the center of the universe. Still faced a similar challenge – he was the sole proponent of his model of medicine, amidst a sea of inflexible thinking and behavior. In an environment of fierce opposition, Still developed a physical model and mechanical approach to the cause and treatment of disease, incorporating minimal use of drugs and surgical intervention. Upon this approach, he founded the American School of Osteopathy in order “to improve our present system of surgery, obstetrics, and treatment of diseases generally” (Still 1897, 168).
Recognizing himself to be a solitary voice raised in opposition to the rigid medical paradigm of his era, and having himself traveled westward in a covered wagon in 1836 (Still 1897, 51), it is not surprising that Still was drawn to the words of Robert Harris, a Kirksville resident who said, “only few men allow themselves to think outside of popular ruts” (Still 1897, 127). This phrase, lamented Still (1897, 127), “. . . was the phrase of all phrases which gave me comfort and support when men rejected the truth and did not accept it.”
W.G. Sutherland was also conscious of the difficulty that large groups have in maintaining flexibility of thought, a phenomenon he referred to as “orthodox grooves” (Sutherland 1962, 13). (Note the similarity of the metaphor to that used by Still.)
Can thinking outside of popular ruts advance our conceptual and practical approach towards the all-pervasive tissue known as fascia? Rut-thinking has certainly been reinforced by how we study fascia, i.e. through anatomy atlases and cadaver dissections. Neither of these mediums offers the “living picture” of anatomy, an image that A.T. Still urged his students to acquire (Still 1899, 13, 42; Still 1902, 9, 89). Atlases offer two-dimensional images with little textual description. Moreover, virtually all the fascia has been omitted in these images in order to reveal the underlying important structures. Cadavers, meanwhile, are completely devoid of fluid.
A four-pronged rut seems to exist with regard to the osteopathic conception of fascia.
1. There is an excessive concentration on the fibrous aspect of fascia, and thus a relative disregard for its other important components – the fluid, the cells, and the matrix – best described together under the term ‘liquid matrix’.
2. There is a preoccupation with its continuity, versus its contiguity.
3. There is a predominant focus on the dense, regular category of connective tissue, rather than the loose connective tissue.
4. The practice of elongation as a modality of release, rather than the providing of spatial separation (and thus fluid flow) between layers.
How was this rut reinforced? Over the last century approximately twenty significant articles on fascia were published in the American osteopathic literature (see Additional Bibliography). They are not experimentally based; instead, they are all anecdotal in nature. When summarized, they fall into four categories:
1. Articles that emphasize the importance that A.T. Still placed upon fascia.
2. Articles that discuss the physical characteristics and properties of the fascial tissue, including its continuity.
3. A majority of articles, describing the anatomical and physiological details of fascial tissue.
4. A few articles specifically discussing fascia’s fluidic or liquid aspect.
Based upon these articles, the osteopathic profession has, as have many other professions, concentrated on the continuous, dense, regular, fibrous composition of fascia, attempting to stretch, release, unwind, manipulate, or normalize it.
Established theory dictates that human collagen fibers align themselves along lines of stress. Applying this information, it seems logical that, if a fascial tissue is assessed as restricted in a particular direction, this indicates that the tissue has been subjected to too much force in that direction. Treatment is usually focused on addressing the origin of the excessive force, the effects of the force (i.e., the restricted nature of the fascial tissue), or both.
Realistically, however, connective tissue, including fascia, is too intricate a tissular system to operate under the simple premise that mechanical stress causes collagen fibers to align themselves; this statement oversimplifies the entire process. In fact, the very existence of fascial tissue represents ongoing interactions and reactions among nonlinear complex sets and subsets of systems and environments, processes that began operating shortly after conception and that continue operating, even while the patient is lying on your treatment table. Fascia exists as the dynamic interaction of its constituents (water, fibers, matrix, and cells) and its environment, which, when combined together, create a tissue that is greater than the sum of its components.
Fascia responds to a whole array of internal and external environmental stimuli, including pressure gradients, tension fields, nutrient (inorganic and organic) availability (in the liquid matrix) and assimilation, vascularization, as well as neural stimulation or lack thereof. It reacts to the addition and withdrawal of mechanical, chemical, thermal, pharmacological, electromagnetic, and even emotional stimuli. Finally, if water is absent, the only sure reaction is cellular death; none of fascia’s purposeful reactions can take place, including the production and arrangement of its fibers.
The biochemical reactions between water and phospholipids which serve to reinforce the integrity of cellular membranes must also be considered. Fascia’s thixotropic nature is a product of the constant interaction of water with the macromolecules (proteoglycans and glycoproteins) of the matrix. [Some osteopaths have even hypothesized that this process is necessary for the operation of the primary respiratory mechanism (Lee 2001, Gabarel and Roques 1985)]. Most of the body’s biochemical activities, including growth, tissue repair, cellular respiration, hormone production and transport, the immune response, the inflammatory response, the resorption of metabolic waste products, as well as every neural impulse, require a fluid medium for their operation.
The above enumeration of fluid function highlights the significance of the rehydrating nature of fluid, as it reaches areas of compromised fascial tissue, including not only the fibers, but also the liquid matrix. In light of all this information, why then are practitioners still trying to release fascia and other aponeurotic tissues through mechanical methods primarily focused on the fibers, methods that sometimes cause the wringing out of the fluids?
Scientific evidence indicates that the tensile strength of human fascia lata tissue was recorded in 1931 as 7,000 pounds per square inch. It was found that the fascia could be elongated by loading it, but that it quickly returned to its resting length once unloaded (Gratz 1931). More recently, crural fascia from five unembalmed human limbs was examined (Stecco et al. 2009). The researchers reported that the mean thickness of the crural fascia was 924?m; that it was composed of two to three layers of “collagen tissue bundles,” each having a mean thickness of 277.6?m; that each of these layers was separated by a layer of loose connective tissue with a mean thickness of 43?m – a thickness undetectable by the unaided human eye. Finally, the researchers reported that the fibers of each layer of the crural fascia were oriented at a different angle from those of the previous layer.
While fascia is indeed continuous, a seldom examined property is its contiguity. While the term ‘continuity’ is usually reserved for the end-to-end linking of tissues, ‘contiguity’ refers to the overlapping of tissue layers in an interior to exterior (deep to superficial) orientation, or vice versa. On repeated occasions this author has had the opportunity to dissect the tissue of freshly (one to four hours) killed deer. When examined visually, the complex nature of the contiguous fibrous layering is immediately apparent. In fact, the contiguous layering of connective tissue in the region of the thigh, unlike the previously given example of the crural fascia in humans, appears to be composed not of two to three, but of almost countless, layers.
These contiguous layers can be easily teased apart using a scalpel. Some layers are so thin that when a single layer is lifted from its underlying layer, it appears to break apart, losing its integrity like the film of a soap bubble (see Figure 3). Of even greater significance is the nature of the connections between overlapping layers: similar to the above findings on crural fascia, the contiguity of these layers is reinforced by loose connective tissue. Thus, as one fibrous layer is lifted off from its underlying layer, a stringy mass of fibers resembling Christmas angel hair is exposed – the loose connective tissue (see Figure 4).

Figure 3: A single layer of fascial tissue from a deer, three hours after death. Photo © Jane Stark, 2009, used with permission.
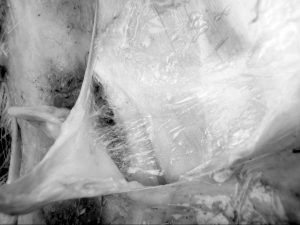
Figure 4: The ‘angel hair’ look of the fibers in the loose connective tissue layer; fascial tissue from a deer, not long after death. Photo © Jane Stark, 2009, used with permission.
Unlike angel hair, which is dry, loose connective tissue is primarily a liquid matrix. It is permeated with fluid due to the water-attracting and water-binding properties of its macromolecules (see Figure 5). Unfortunately, confirming this observation is very difficult because the moment the layers are separated, the tissue dehydrates. Gas bubbles appear as the integrity of the tissue layers is mechanically disrupted, opening a previously closed system and altering pressures (see Figure 6).
However, from this author’s personal observations, when two contiguous layers are left unseparated, their combination of fluid and fibers remains intact and can be investigated. The arrangement appears to have a twofold mechanical purpose. It allows the superficial layer to glide very easily (through a confined range) over its underlying layer. The fluidic aspect allows the gliding, while the fibers delimit its range and direction of excursion.
Two observations arise upon further examination of this contiguous layering: 1) the more superficial the layer, the more excursion it has; and 2) each layer appears to have a preferred direction of excursion, one that is usually different from that of the layer underlying it. How then is it possible to elongate a unit of tissue, such as the iliotibial band, if it is composed of numerous layers, each of which appears to have a differing preferred direction of permitted motion?
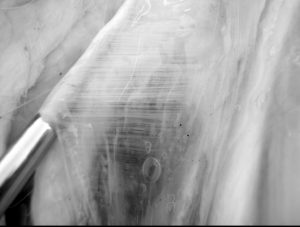

Figure 5. The fluid environment of dense connective tissue (in a deer, shortly after death). Photos © Jane Stark, 2008, used with permission.
Having revisited all these details of the complex nature and operation of fascial tissue, it may now be practical to add to the accepted view of fascia as a continuous layer of dense fibrous connective tissue – the perspective of fascia as liquid matrix. It is possible now to re-examine elongation methods in light of the idea that they may actually serve to wring out fluid, instead of facilitating its uptake. By combining scientific research with observation of fresh tissue it is surely possible to produce a more productive and appropriate alternative to the purely fibrous approach. This new approach would include the drawing or attracting of fluid toward loose connective tissue, with the intent of suspending each contiguous fibrous layer of dense tissue within the liquid matrix environment provided by the spaces within loose connective tissue. Is the idea of concentrating on the fluidic nature of fascia new? No! Sutherland suggested it in 1953:
• “The fascia! Even the fascia is water, even the bony tissue is liquid . . . water . . . fluid . . . Fluid!” (Sutherland 1953, 290)
• “. . . You will find space between [the lines of the fascia] if you have the vision to look in between. A microscope powerful enough to see the space between.” (Sutherland 1953, 295)
• “When the tide comes in and the waves roll over that rock [comparing the spaces within fascia to the spaces between the layers of a rock], you will find it [the rock] crumbling into sand . . . Sand!” (Sutherland 1953, 295)
Sutherland’s ideas no longer seem to be reflected in the fascial treatment practices of currently practicing osteopaths. Qualitative research conducted in 2001- 2003 demonstrated that an international sample of experienced osteopaths and osteopathic physicians, having a total of over 1,200 years of clinical practice between them, did not seem to consider the fluid aspect in the treatment of fascia, even though several identified fascia as having a fluidic or flowing or liquid aspect to it when in a healthy state.
In personal interviews with thirty-six osteopathic practitioners (Stark 2007, 194- 198), each having twenty or more years of experience in a manual-based practice, only a few discussed the fluid aspect when asked, “How do you know when your fascial treatment is successful?” While some dismissed the idea of being able to separate fascia from any other tissue and, thereby, any attempt to treat it individually, those who did discuss specific fascial treatment mostly judged their success by observing less tension and more symmetry, motion, mobility, and motility. There only two or three (of thirty-six) mentions of changes in fluidity, the quality of the liquid matrix, circulation, nervous activity, or lymphatic status as indicators of a successful fascial treatment (Stark 2007, 206-209, 346-348). The most fitting response to the question “How do you know when your fascial treatment is successful?” (Stark 2007, 194- 198) was, “You know when the fascia has imbibed fluids the quality of the fascia has changed because it has recovered fluid quality” (Stark 2007, 347). Also emphasizing the fluidic importance of fascia is a 2006 article entitled “The Effects of Manipulation on Ligaments and Fascia from a Fluids Model Perspective.” The author, Thomas Crow, DO, FAAO wrote, “When you feel the flow come through the dysfunctional area, your treatment of that area is complete” (Crow 2006).
These two statements certainly reinforce the salient point of this article, which is to appreciate the loose connective tissue which supports the contiguity of the dense fibrous tissue layering of fascia, its fluid aspect, and to consider the fibers as residing within a liquid-matrix environment upon which their integrity, longevity, and usefulness depends.
Investigating and acknowledging our own rut-like thinking is a difficult exercise, potentially as overwhelming as coming to terms with a round planet Earth after centuries of knowing it to be flat. Yet such paradigm shifts have been accomplished in osteopathy, beginning with A.T. Still. This review offers us the perfect forum in which to think outside of popular ruts and to revisit our understanding of fascia from the perspective of a liquid matrix.
Jane Eliza Stark is a 2003 graduate of the Canadian College of Osteopathy (CCO), where upon graduation she received the Andrew Taylor Still Award for the most representative thesis on the advancement of osteopathy in philosophical or sociological research. In 2006, she received the Andrew Taylor Still Foundation award for her contribution to the advancement and recognition of osteopathy in Canada. She serves on the faculty of the CCO and the Collège D’Etudes Ostéopathiques. She has a master’s degree in clinical research administration and is the director of research for the CCO. She is recognized internationally as an osteopathic historiographer, author, and lecturer on osteopathic history and on the biographies of Still, Sutherland, and Littlejohn. She is a workshop leader on the fluidic approach to treating connective tissue and has lectured or taught in ten countries. She will be giving a seminar on the History of Osteopathy in November in Toronto; see http://foundersdayweekend.ca/Seminars/ Seminars%20Description/ for information.
Bibliography
Crow, T. 2006. “The Effects of Manipulation on Ligaments and Fascia from a Fluids Model Perspective.” The AAO [American Academy of Osteopathy] Journal September 2006:13-19.
Gabarel, B. and M. Roques 1985. Les Fasciae en Médecine Ostéopathique. Paris: Maloine.
Gratz, C.M. 1931. “Tensile Strength and Elasticity Tests on Human Fascia Lata.” The Journal of Bone & Joint Surgery 13:334-340.
Lee, R.P. 2001. “The Primary Respiratory Mechanism Beyond the Craniospinal Axis.” American Academy of Osteopathy Journal 11(1):24-34.
Stark, J.E. 2007. Still’s Fascia: A Qualitative Investigation to Enrich the Meaning Behind Andrew Taylor Still’s Concept of Fascia. Pähl, Germany: Jolandos Verlag. [Editor’s note: Jolandos Verlag is continuing to print a German edition of this book; an English edition should be forthcoming in the U.S., please contact [email protected] for more information if interested.]
Stecco, C., A. Porzionata, A. Stecco, and R. De Caro 2009. “Anatomical Study and Tridimensional Model of the Crural Fascia.” Paper presented at the Second International Fascia Research Conference, Vrije Universiteit, Amsterdam.
Still, A.T. 1897. Autobiography of Andrew Taylor Still with a History of the Discovery and Development of the Science of Osteopathy. Kirksville Mo: self-published by A.T. Still.
Still, A.T. 1899. Philosophy of Osteopathy. Kirksville, Missouri: self-published by A.T. Still.
Still, A.T. 1902. The Philosophy and Mechanical Principles of Osteopathy. Kansas City, Missouri: Hudson-Kimberly Publishing Company.
Sutherland, A.S. 1962. With Thinking Fingers: The Story of William Garner Sutherland. Kansas City, Missouri: The Cranial Academy.
Sutherland, W. 1953 (January 9). “Liquid Light.” Included in Contributions of Thought: The Collected Writings of William Garner Sutherland, D.O., Pertaining to the Art and Science of Osteopathy Including the Cranial Concept in Osteopathy Covering the Years 1914-1954, A.S. Sutherland and A.L. Wales, Foundation SCT, Eds., Portland, Oregon: Rudra Press, 1971/1998, pp. 289-296.
Additional Bibliography
Significant Articles on Fascia in the American Osteopathic Literature of the Past Century (in reverse chronological order)
Crow, T. 2006. “The Effects of Manipulation on Ligaments and Fascia from a Fluids Model Perspective.” The AAO [American Academy of Osteopathy] Journal September 2006:13-19.
Typaldos, S. 1994. “Introducing the Fascial Distortion Model.” AAO Journal 4:14-18, 30-36.
Becker, F. 1975 (Feb). “The Meaning of Fascial and Fascial Continuity.” Osteopathic Annals 3(2):8-32.
Greenman, P.E. 1975 (Feb). “Fascial Considerations in Treatment of the Head and Neck.” Osteopathic Annals 3(2):34-42.
Cathie, A. 1974. “Consideration of Fascia and its Relation to Disease of the Musculoskeletal System.” AAO Yearbook 85-88.
Cathie, A. 1974. “Fascia of the Body in Relation to Function and Manipulative Therapy.” AAO Yearbook 81-84.
Magoun, H.I. 1970. “Fascia in the Writings of A.T. Still.” AAO Yearbook 159-168.
Snyder, G. 1970. “Embryology and Physiology of Fascia.” AAO Yearbook 147- 158.
“PCOM Class of 1969” 1968. “The Clinical Importance of Fascia,” A. Cathie and R.W. England, Eds. AAO Yearbook Selected Papers 1968: 87-103
Erlingheuser, R. 1959. “The Circulation of the Cerebrospinal Fluid Through the Connective Tissue System.” AAO Yearbook 77-87.
Snyder, G. 1956. “Fasciae – Applied Anatomy and Physiology.” AAO Yearbook 65-75.
Hitch, S. 1954. “Malalignment of the Sternum and Fascia Strain.” Journal of the Osteopathic Cranial Association 1954: 23-30.
Page, L.E. 1952. “The Role of the Fasciae in the Maintenance of Structural Integrity.” AAO Yearbook 70-73.
Hoover, M.A. 1951. “Importance of Fascia.” AAO [American Academy of Osteopathy] Yearbook 55-72.
Arbuckle, B.E. 1947 “Reflexes.” Journal of the American Osteopathic Association 46(7):405- 407.
Becker, A.R. 1947 (Feb). “The Fascias.” Osteopathic Profession XIV(5):13-17, 36, 38.
Denslow, J.S. 1947 (Feb). “Soft Tissues in Areas of Osteopathic Lesion.” Journal of the American Osteopathic Association 46(6):334- 337.
Kauffman, C.H. 1944. “Physiologic Basis of A.T. Still Technic.” The Osteopathic Profession 12(2):17- 20, 48, 50, 52.
McConnell, C.P. 1939 (Dec). “Fundamental Fragments: IV Fascia.” Journal of the American Osteopathic Association 39(4):204.
Kerr, H.E. 1936 (May). “The Role of Fascia in Osteopathic Problems.” The Journal of the American Osteopathic Association 35(9):418- 420.
McConnell, C.P. 1928. “Studies in Osteopathic Pathology.” The Journal of the American Osteopathic Association 28(3):167- 170.
Millard, F.P. 1923 (Mar). “Fascia-TensionLight.” The Journal of the American Osteopathic Association 22(7):402, 405-406, 417-418.

To have full access to the content of this article you need to be registered on the site. Sign up or Register.